LINK® Embrace Shoulder System - Reverse Configuration
Shoulder Prosthesis, Reverse Configuration
Waldemar Link GmbH & Co. KG
The following data is part of a premarket notification filed by Waldemar Link Gmbh & Co. Kg with the FDA for Link® Embrace Shoulder System - Reverse Configuration.
Pre-market Notification Details
| Device ID | K212992 |
| 510k Number | K212992 |
| Device Name: | LINK® Embrace Shoulder System - Reverse Configuration |
| Classification | Shoulder Prosthesis, Reverse Configuration |
| Applicant | Waldemar Link GmbH & Co. KG Ostsraße 4-10 Norderstedt, DE 22844 |
| Contact | Stefanie Fuchs |
| Correspondent | Terry Powell LinkBio Corp 69 King Street Dover, NJ 07801 |
| Product Code | PHX |
| Subsequent Product Code | HSD |
| Subsequent Product Code | PAO |
| CFR Regulation Number | 888.3660 [🔎] |
| Decision | Substantially Equivalent (SESE) |
| Type | Traditional |
| 3rd Party Reviewed | No |
| Combination Product | No |
| Date Received | 2021-09-20 |
| Decision Date | 2021-11-18 |
Trademark Results [LINK]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 LINK 98507530 not registered Live/Pending |
Stripe, Inc. 2024-04-18 |
 LINK 98450996 not registered Live/Pending |
Smart Tracking Technologies, LLC 2024-03-15 |
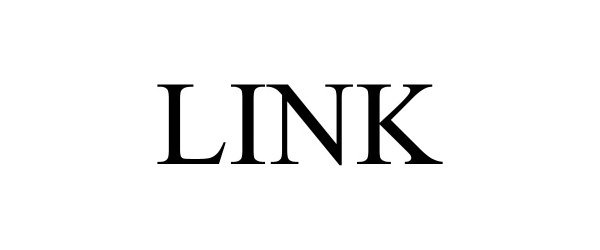 LINK 98157539 not registered Live/Pending |
Pradopark, S.A. 2023-08-30 |
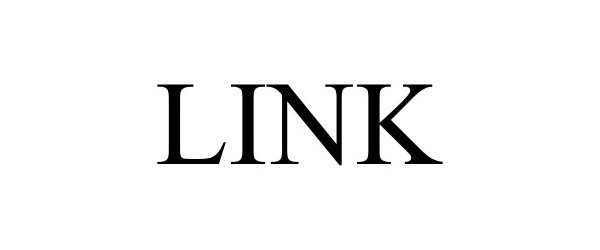 LINK 98090489 not registered Live/Pending |
REV Recreation Group, Inc. 2023-07-18 |
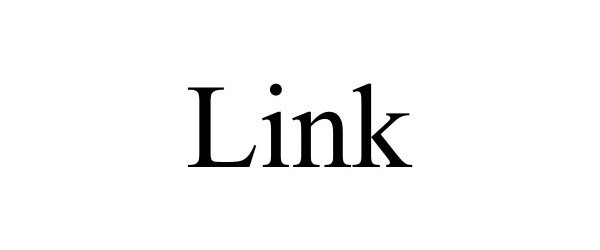 LINK 98031955 not registered Live/Pending |
Liminal Strategy Partners LLC 2023-06-07 |
 LINK 97873463 not registered Live/Pending |
Intrinsic Group Limited 2023-04-05 |
 LINK 97705709 not registered Live/Pending |
LINK S.R.L. 2022-12-06 |
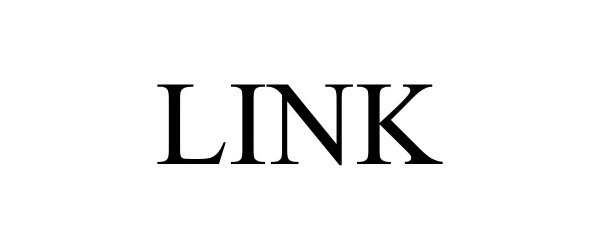 LINK 97682049 not registered Live/Pending |
Sxanpro, LLC 2022-11-17 |
 LINK 97653413 not registered Live/Pending |
Huynh, Phat Huu 2022-10-28 |
 LINK 97651668 not registered Live/Pending |
Pradopark, S.A. 2022-10-27 |
 LINK 97521122 not registered Live/Pending |
Link Brokerages, Inc. 2022-07-26 |
 LINK 97364999 not registered Live/Pending |
Link Mfg Ltd. 2022-04-15 |
© 2024 FDA.report
This site is not affiliated with or endorsed by the FDA.